In Action
Our Benefits
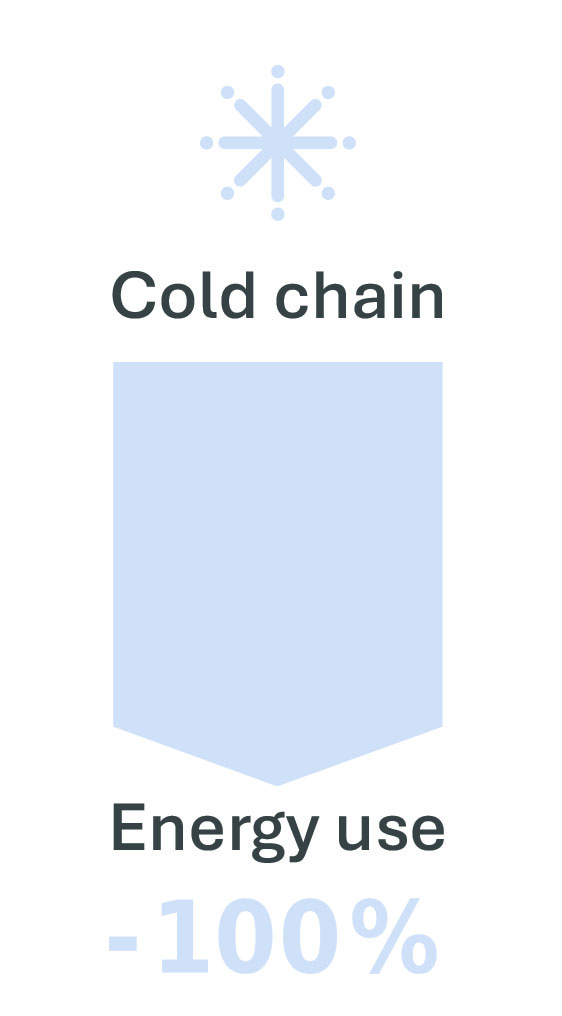
Manufacturing & Shipping significantly reduces the carbon footprint vs Needle & Syringe
Vaccines – Providing a much Cleaner Next-Gen Technology
Supported by data from a PATH Vaccine Technology Impact Assessment



Shipping Economy & Logistics Efficiency – Containers

Up to 1.3 Million aVaxziPen doses
In a Standard 20ft Shipping Container

Up to 220 Thousand liquid N&S doses*
In a Medical Grade Cold Chain 20ft Shipping container
Up to 6 times more aVaxziPen vaccinations in the same size container vs N&S provides huge shipping & logistics economy
*For a reconstituted single-dose vial vaccine. (Both container quantities assume standard 1.2m x 1.2m x 1m pallets stacked floor to ceiling)

